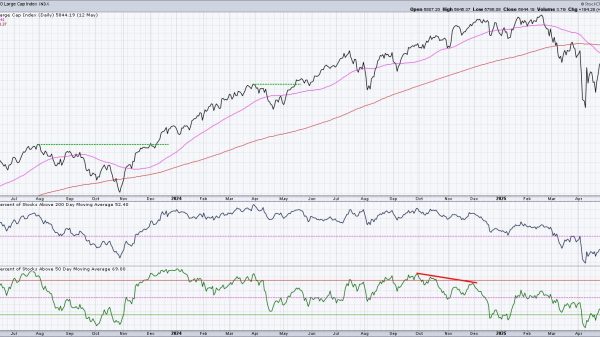
The FDA’s 16-member advisory panel unanimously voted yesterday that oral phenylephrine, a common active ingredient in cold medications, is no better than a placebo for treating congestion. The call by the panel sets up potential FDA action that could force the removal of certain over-the-counter medications containing the ingredient — including certain formulations of Mucinex, Sudafed, Tylenol, and NyQuil — from store shelves.
As noted by The New York Times, the panel’s vote doesn’t necessarily mean the FDA will take action, but it usually follows panel recommendations. That said, the FDA may hold off for many months, pending contested findings by drug makers and other considerations.
The panel tossed out several older studies that…
Continue reading…